马上注册,阅读更多内容,享用更多功能!
您需要 登录 才可以下载或查看,没有账号?立即注册 
×
随着液体活检技术发展,通过血液检测ctDNA来发现微小残留病(MRD),已成为早期肺癌术后复发监测的重要工具。但目前检测平台、时机和结果解读缺乏统一标准。本研究采用严谨的德尔菲法(Delphi方法),由亚洲胸部肿瘤学研究组(ATORG)组织27位专家,通过两轮匿名投票形成共识(≥80%同意即为强共识)。最终形成23条共识声明,其中22条达成强共识,为早期肺癌MRD检测提供了统一的临床应用框架。


Figure 1. Consensus levels of position statements across thematic domains on MRD testing in early-stage NSCLC developed through Delphi methodology.
PART.01 Assay Validity and Standardization
1.1
The minimum analytical sensitivity threshold for MRD assays in early-stage resected NSCLC, based on currently available technologies, should be defined and adopted.
基于现有技术,应定义并采纳早期可切除NSCLC中MRD检测的最低分析灵敏度阈值。 1.2
Publications of MRD assays should be recommended to report key metrics including variant allele frequency (VAF) for tumor-informed assays if available, limit of detection (LOD), sensitivity and specificity.
建议MRD检测的相关文献应报告关键指标,包括(如适用)肿瘤知情检测方法的等位基因突变频率、检测下限、灵敏度和特异性。 1.3
Cross-platform benchmarking frameworks should be developed to facilitate valid comparison of analytical performance and result interpretation across both tumor-informed and tumor-agnostic MRD assays.
应建立跨平台基准比较框架,以促进肿瘤知情和肿瘤不知情MRD检测方法之间的分析性能与结果解读的有效比较。 1.4
Guidelines for pre-analytical handling (e.g. sample type, storage time, plasma volumes) to standardize MRD sample collection should be developed and circulated.
应制定并推广关于前分析处理(如样本类型、保存时间、血浆体积)的指南,以规范MRD样本采集。
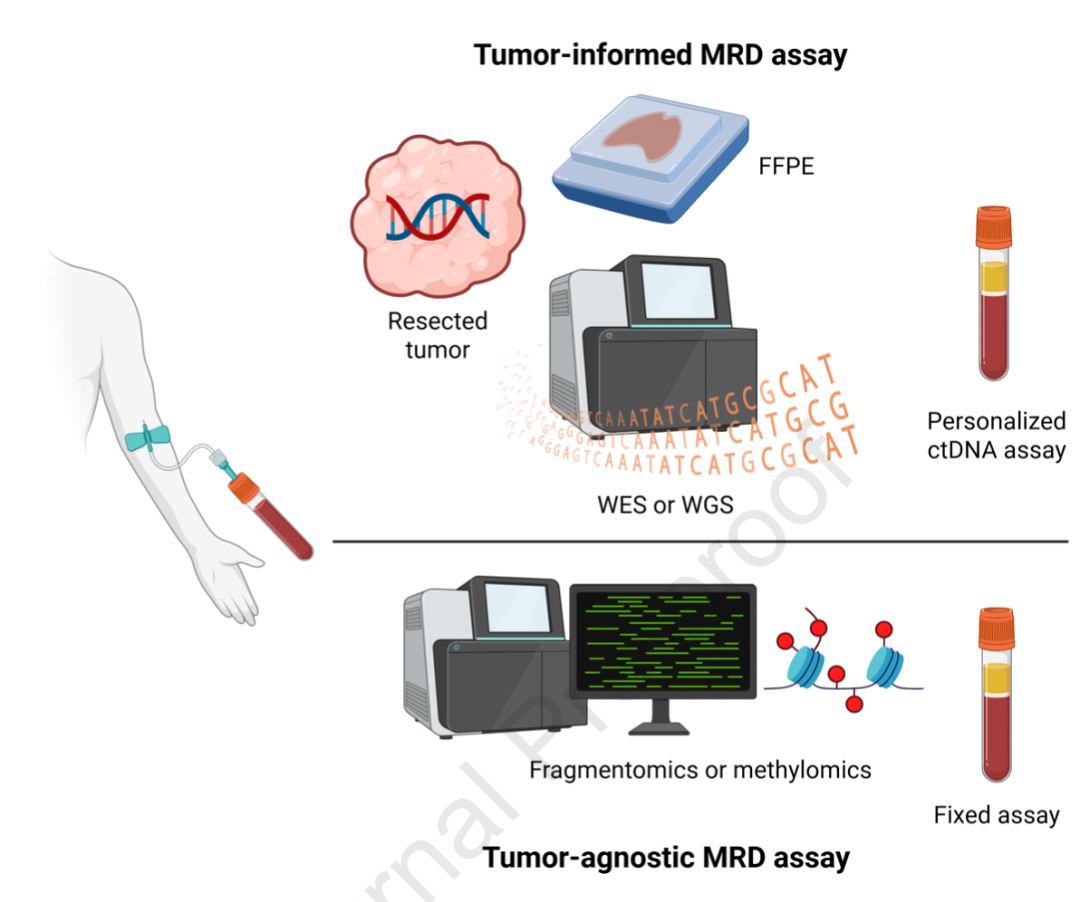
Figure 2. Overview of tumor-informed and tumor-agnostic assays for MRD testing in early- stage NSCLC
PART.02 Harmonization in Research and Trials
2.1
Clinical research and trials involving MRD in early-stage NSCLC should harmonize timing and terminology of blood sampling: (1) For an upfront surgery clinical trial, blood sampling timepoints should be aligned to at least baseline, post-surgery MRD landmark, the completion of adjuvant therapy and longitudinal follow up. (2) For a perioperative clinical trial, blood sampling timepoints should be aligned to at least baseline, completion of neoadjuvant therapy, post-surgery MRD landmark, the completion of adjuvant therapy and longitudinal follow up.
涉及早期NSCLC中MRD的临床研究和试验应统一采血时间点与术语:(1)对于直接手术的临床试验,采血时间点至少应包括基线、术后MRD评估点、辅助治疗结束后和长期随访;(2)对于围手术期临床试验,采血时间点至少应包括基线、新辅助治疗结束后、术后MRD评估点、辅助治疗结束后和长期随访。 2.2
By harmonizing timing and terminology of blood-based tests in research and trials for early-stage NSCLC, we could confirm the prognostic value of baseline blood-based testing results, clearance of blood-based markers after completion of neoadjuvant therapy and post-surgery MRD landmark.
通过统一早期NSCLC研究和试验中血液检测的时间点与术语,可以确认基线血液检测结果、新辅助治疗结束后及术后MRD评估点的血液标志物清除情况的预后价值。 2.3
By harmonizing timing and terminology of blood-based tests in research and trials for early-stage NSCLC, we could build evidence for use of post-surgery MRD landmark as guidance for post-surgical adjuvant therapy escalation/de-escalation.
通过统一早期NSCLC研究和试验中血液检测的时间点与术语,可以为术后MRD评估点作为辅助治疗升级/降级依据建立证据。 2.4
By harmonizing timing and terminology of blood-based tests in research and trials for early-stage NSCLC, we could build evidence for optimization of longitudinal monitoring of cancer status.
通过统一早期NSCLC研究和试验中血液检测的时间点与术语,可以为优化肿瘤状态的纵向监测建立证据。
PART.03 Clinical Application
3.1
Current MRD testing assays demonstrate high specificity and positive predictive value but variable sensitivity and negative predictive value for predicting recurrence after surgical resection. An undetectable MRD result does not rule out recurrence.
当前MRD检测方法在预测术后复发方面具有较高的特异性和阳性预测值,但灵敏度和阴性预测值存在差异。MRD检测不到并不排除复发可能。 3.2
Due to the limited sensitivity of current MRD detection methods, an undetectable MRD result after surgical resection should not yet be used to guide adjuvant therapy de-escalation in clinical practice until further technological improvements are achieved.
由于当前MRD检测方法的灵敏度有限,在技术进一步改进之前,术后MRD检测不到的结果尚不应用于临床实践中指导辅助治疗降级。 3.3
A detectable MRD after surgical resection is a strong negative prognostic biomarker; however, there is a lack of robust prospective evidence for using MRD to guide specific therapeutic recommendations.
术后检测到MRD是一个强阴性预后生物标志物;然而,目前缺乏强有力的前瞻性证据支持使用MRD来指导具体的治疗建议。
PART.04 Challenges in Implementation
4.1
Access to MRD testing is encouraged - whether within the country or neighboring countries - to support the generation of local/regional evidence.
鼓励在本国或邻国获取MRD检测,以支持本地/区域性证据的生成。 4.2
Funding pathways or grant mechanisms in each country are recommended to support the integration of MRD assays into trials.
建议各国建立资金渠道或资助机制,以支持将MRD检测整合到试验中。 4.3
Value of MRD testing needs to be balanced against competing healthcare priorities, including improving access to established imaging, pathology, systemic therapy, and radiotherapy.
MRD检测的价值需要与其他竞争的医疗优先事项(包括改善影像、病理、系统治疗和放射治疗的可及性)进行平衡。 4.4
In the absence of universally accepted guidelines, MRD testing in lung cancer should remain largely within clinical trial settings until there is evidence on how results can influence adjuvant or surveillance strategies.
在缺乏普遍接受的指南的情况下,肺癌的MRD检测应主要限于临床试验环境,直到有证据表明其结果可以影响辅助治疗或监测策略。
PART.05 Consensus Recommendations
5.1
An MRD consensus working group should be convened, to regularly update international guidelines for lung cancer based on emerging evidence.
应召集MRD共识工作组,根据新出现的证据定期更新肺癌国际指南。 5.2
The use of ctDNA for MRD assessment should be supported by rigorous validation (e.g. radiologic and clinical correlates) in ongoing and completed adjuvant, neoadjuvant, and peri-operative therapy trials.
在正在进行和已完成的辅助、新辅助及围手术期治疗试验中,使用ctDNA进行MRD评估应得到严格的验证(如影像学和临床相关性)支持。
PART.06 Infrastructure for Regional MRD Adoptions
6.1
Regional clinical practice guidelines for MRD testing in early-stage NSCLC should be developed to enable evidence-based adoption.
应制定针对早期NSCLC的MRD检测区域临床实践指南,以促进基于证据的推广应用。 6.2
Clinician- and patient-education helps to support appropriate use of MRD.
临床医生和患者教育有助于支持MRD的合理使用。 6.3
Collaborative collection of real-world MRD data across Asia- PACIFIC countries is needed to generate evidence that informs research, reimbursement, and policy.
需要在亚太各国之间合作收集真实世界的MRD数据,以生成能够指导研究、报销和政策制定的证据。
PART.07 Roadmap for Pragmatic Trials
7.1
Given its strong prognostic value, MRD is a valuable tool for patient stratification. Prospective clinical trials investigating both therapy escalation (in MRD-positive patients) and de-escalation (in MRD-negative patients) are recommended.
鉴于其强大的预后价值,MRD是一个有价值的患者分层工具。建议开展前瞻性临床试验,同时研究治疗升级(针对MRD阳性患者)和治疗降级(针对MRD阴性患者)。 7.2
MRD-guided treatment strategies aim to improve outcomes by escalating therapy in MRD-positive patients, where intensification may enhance survival, and by de-escalating in MRD-negative patients to reduce treatment burden without compromising effectiveness.
MRD指导的治疗策略旨在通过以下方式改善预后:对MRD阳性患者升级治疗(强化可能提高生存率),对MRD阴性患者降级治疗(在不影响疗效的前提下减少治疗负担)。 7.3
When designing perioperative MRD-guided clinical trials, it is critical to consider the distinct biology and standard of care associated with different driver genes. Stratifying trial populations by driver gene status — particularly separating EGFR/ALK-altered from wild-type patients — is a key consideration.
在设计围手术期MRD指导的临床试验时,必须考虑不同驱动基因相关的独特生物学特性和标准治疗方案。根据驱动基因状态对试验人群进行分层——特别是将EGFR/ALK突变患者与野生型患者分开——是一个关键考虑因素。
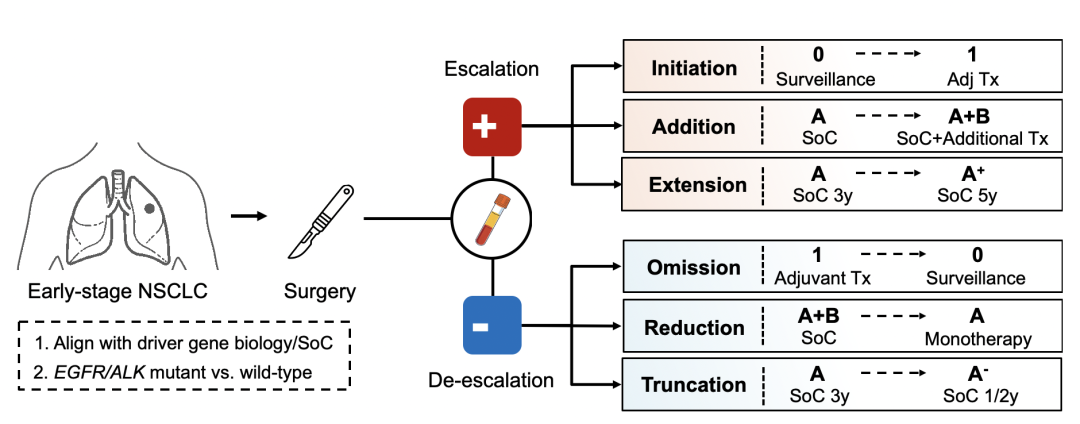
Figure 3. Proposed roadmap for MRD-guided clinical trials in early-stage NSCLC
总结:这份由ATORG发布的共识声明为ctDNA-based MRD检测在早期NSCLC中的临床和研究应用提供了一个全面、由专家主导的框架。专家组达成了强烈共识,即MRD是一个重要的预后生物标志物,但尚不能作为常规临床应用中的确定性预测工具。关键的是,MRD检测不到的结果并不能可靠地排除复发,因此临床解读需要谨慎。检测方法的标准化和统一(包括前分析变量)仍然是临床推广的重要前提,这将有助于跨平台基准比较以及不同研究之间的结果对照,而目前研究结果的可推广性仍然有限。统一的MRD检测时间点和框架也是加速证据积累所必需的。重要的是,目前正在进行的、基于MRD指导治疗升级和降级的试验结果备受期待。未来试验的务实试验设计将使其在真实临床实践中产生更大的影响。此外,为了公平推广,需要在资源有限的环境中建设适当的基础设施,以便能够进行MRD检测。总的来说,这些共识推荐为负责任地将MRD检测整合到临床研究和实践中提供了一个务实的指导框架。
来源文献:
Tan AC, Liao BC, Li M, Lee D, Uehara Y, Thamlikitkul L, Zhang JT, Zheng M, Lee CK, Pavlakis N, John T, Soo RA, Skanderup A, Voon PJ, Ahn BC, Park S, Hayashi H, Goto Y, Horinouchi H, Yatabe Y, Reungwetwattana T, Chih-Hsin Yang J, Kim DW, Mok T, Tan DSW, Wu YL, Ahn MJ. Consensus statement on ctDNA minimal residual disease (MRD) testing in early-stage NSCLC - A Delphi study by the Asian Thoracic Oncology Research Group (ATORG). J Thorac Oncol. 2026 Mar 26:103696. doi: 10.1016/j.jtho.2026.103696. Epub ahead of print. PMID: 41903701.
|