马上注册,阅读更多内容,享用更多功能!
您需要 登录 才可以下载或查看,没有账号?立即注册 
×
宗艾替尼(Zongertinib,BI 1810631)是一种在研的口服HER2特异性酪氨酸激酶抑制剂(TKI),是一种HER2突变非小细胞肺癌(NSCLC)的潜在治疗药物。Zongertinib于2023年获得FDA快速通道资格认定,然后于2024年获得美国FDA和中国CDE的突破性疗法认定,用于治疗具有活化HER2突变且既往接受过全身治疗的晚期NSCLC成人患者。 Beamion LUNG-1研究:是一项正在进行中的Ⅰa/b期试验,旨在评估Zongertinib在HER2阳性实体瘤(Ia期)和HER2阳性NSCLC(Ib期)患者中的安全性和有效性。Ⅰb期总共分为5个治疗队列,2024WCLC会议报告了Beamion LUNG-1研究Ⅰb期队列1的结果。在队列1中,经治HER2酪氨酸激酶结构域(TKD)突变NSCLC患者接受每日120mg或240mg的Zongertinib治疗。主要终点为中央独立盲态审查(BICR)确认的客观缓解率(ORR);次要终点包括缓解持续时间(DoR)和无进展生存期(PFS)。
 研究结果:截至2024年5月,共有132名患者接受了每日120mg或240mg的Zongertinib治疗(n=75/n=57)。在中央独立盲态审查(BICR)评估下,确证客观缓解率(ORR)为66.7%,97.5%CI(53.8–77.5), (p<0.0001),队列1(120mg;n=75)的主要终点达成。根据研究者评估,所有剂量组中94%的患者观察到了不同程度的肿瘤缩小。试验设计包括剂量递增,以确定该患者群体中Zongertinib的最佳剂量。患者以1:1的比例随机分配至120mg(n=58)或240mg(n=55)组。在中期无效性分析后,选择了120mg作为在队列1中进一步评估的剂量,并额外招募了17名患者。在1:1随机分组的试验部分,接受Zongertinib每日120mg治疗的患者显示出72.4%的缓解率,接受每日240mg治疗的患者中缓解率为78.2%,且疾病控制率(DCR)分别为95%和100%。
研究结果:截至2024年5月,共有132名患者接受了每日120mg或240mg的Zongertinib治疗(n=75/n=57)。在中央独立盲态审查(BICR)评估下,确证客观缓解率(ORR)为66.7%,97.5%CI(53.8–77.5), (p<0.0001),队列1(120mg;n=75)的主要终点达成。根据研究者评估,所有剂量组中94%的患者观察到了不同程度的肿瘤缩小。试验设计包括剂量递增,以确定该患者群体中Zongertinib的最佳剂量。患者以1:1的比例随机分配至120mg(n=58)或240mg(n=55)组。在中期无效性分析后,选择了120mg作为在队列1中进一步评估的剂量,并额外招募了17名患者。在1:1随机分组的试验部分,接受Zongertinib每日120mg治疗的患者显示出72.4%的缓解率,接受每日240mg治疗的患者中缓解率为78.2%,且疾病控制率(DCR)分别为95%和100%。
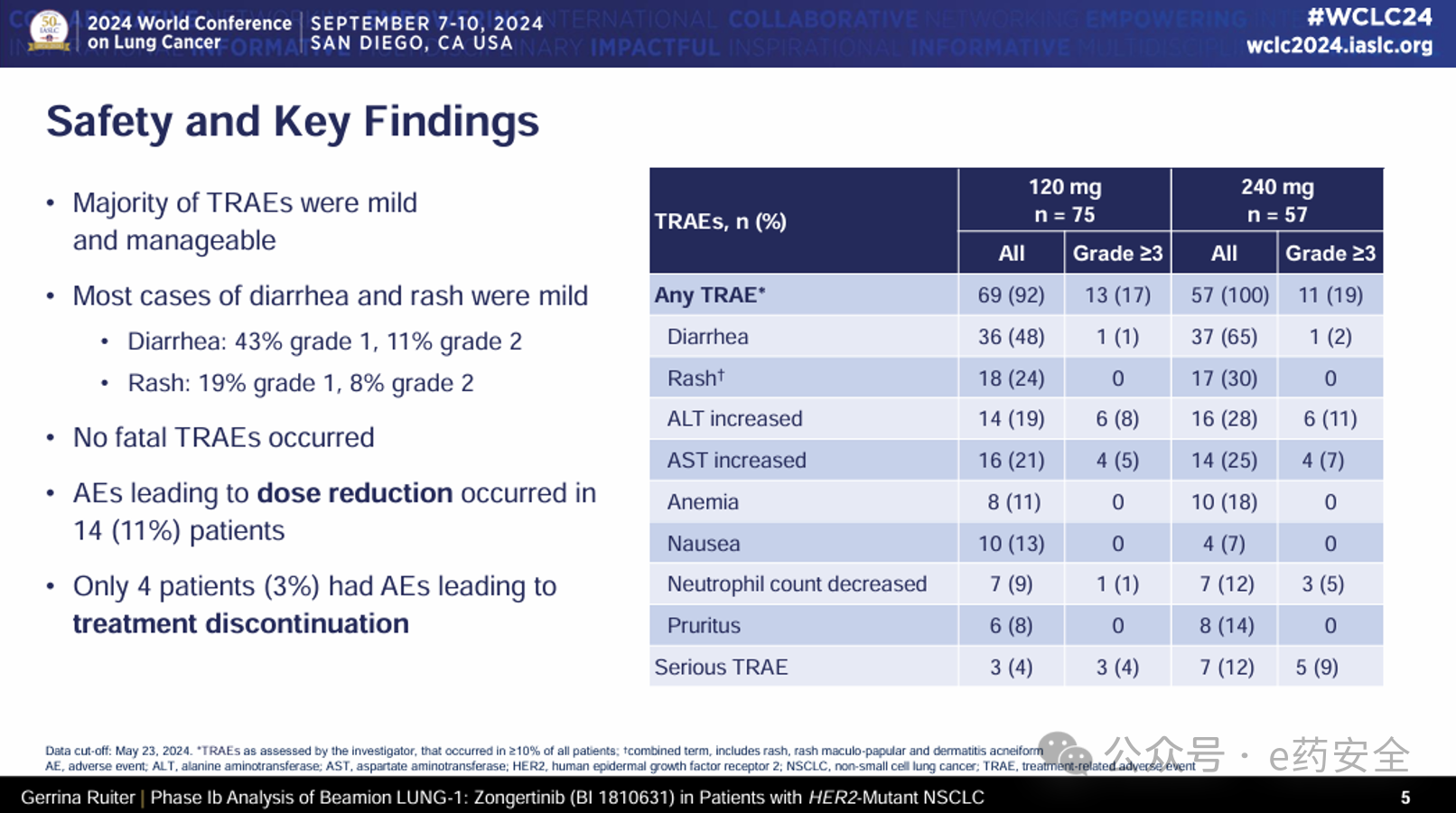
Ib期队列1的数据还初步显示出Zongertinib对脑部病变的活性。根据独立中央盲态审查(BICR)和RANO-BM(神经肿瘤反应评价-脑转移)标准,在无症状脑转移的患者中,33%(120mg;n=27)和40%(240mg;n=25)的患者获得了确认的客观缓解,疾病控制率(DCR)分别为74%和92%。安全性方面,Zongertinib在120mg和240mg剂量下总体耐受性良好,未发生与治疗相关的死亡事件,且导致剂量减少的不良事件发生率(11%)和停药率为(3%)均较低。未观察到新的安全信号或与治疗相关的间质性肺疾病(ILD),3级或以上的治疗相关不良事件(TRAEs)发生率分别为17%(120mg)和19%(240mg)。最常见的治疗相关不良事件为1级或2级腹泻(分别为43%和11%)以及1级或2级皮疹(分别为19%和8%)。
1a/1b期研究结果显示,Zongertinib治疗HER2突变晚期NSCLC的客观缓解率(ORR)达到71%,6个月PFS率为69%,药物安全性良好。
2024 年 ESMO 亚洲大会上公布了 BEAMION LUNG-1 试验 Ib 期队列 1 的最新研究数据,评估 zongertinib(BI 1810631)在治疗携带 HER2 突变阳性晚期 NSCLC 经治患者中的疗效。经中心审查确认,zongertinib 展现出卓越疗效:ORR 为 71%,疾病控制率(DCR)高达 93%。初步生存数据表明,zongertinib 缓解持久:6 个月 PFS 率和 DoR 率分别为 69% 和 73%。在数据截至时,仍有 55% 的患者仍在接受治疗。Zongertinib 作为一种在研分子药物,尚未获得任何医疗监管机构批准,其疗效及安全性仍有待验证,而其所展现的结果,已足以对 HER2 ADC 发起挑战。
宗艾替尼(Zongertinib,BI 1810631)是由勃林格殷格翰公司研发的一款新型、口服的HER2选择性酪氨酸激酶抑制剂,可选择性地与HER2酪氨酸激酶结构域共价结合,包括外显子20插入突变,同时保留野生型表皮生长因子受体。
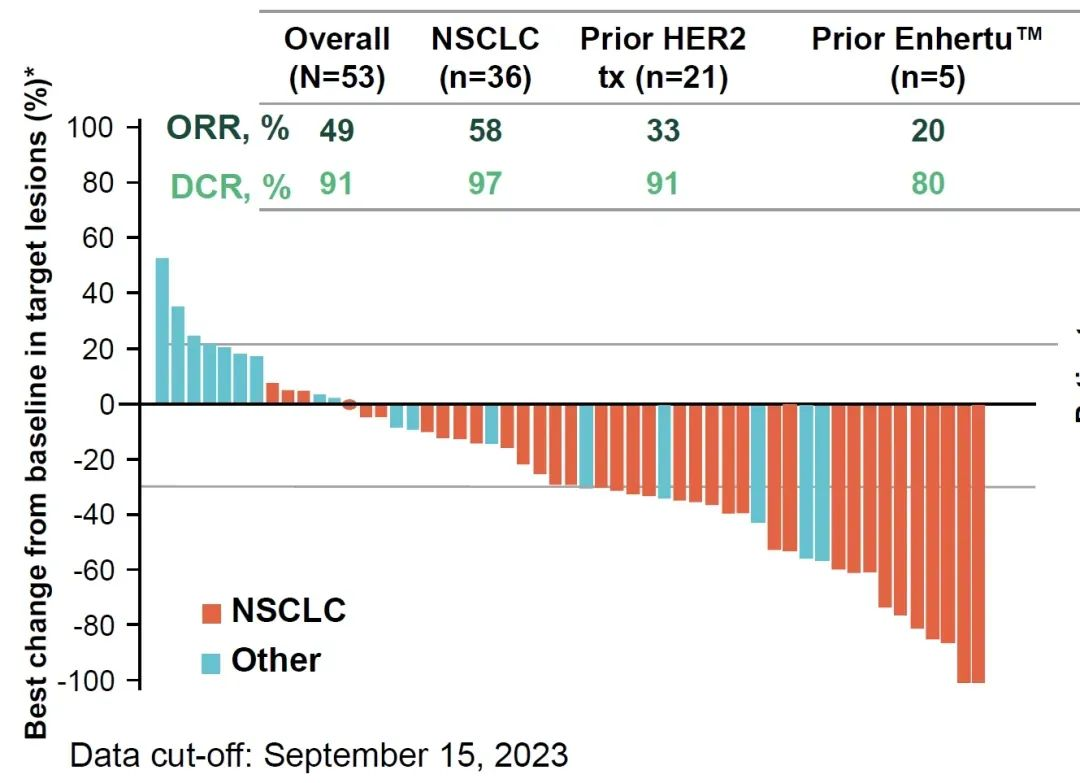
宗艾替尼本次在中国申报上市,主要基于一项Beamion LUNG-1(NCT04886804)Ⅰa/Ⅰb期临床研究的惊艳数据。本次研究共入组61例HER2异常阳性的晚期/不可切除/转移性实体瘤患者,接受剂量递增的宗艾替尼(Zongertinib)口服治疗。
结果显示,中位无进展生存期(PFS)为8.7个月(95%CI:5.5个月~不可评估[NE]);总体中位缓解持续时间为12.7个月(95%CI:4.2~12.7个月)。在53例可评估疗效的患者中,客观缓解率(ORR)达到49%,疾病控制率(DCR)高达91%。
Background:
Zongertinib is an irreversible tyrosine kinase inhibitor that selectively inhibits HER2 while sparing EGFR, thereby limiting associated toxicities. Zongertinib was granted Fast Track and Breakthrough Therapy Designations by the FDA for patients (pts) with advanced, unresectable, or metastatic non-small cell lung cancer (NSCLC) whose tumors have activating HER2 mutations and who have received prior systemic therapy. Beamion LUNG-1 (NCT04886804) is a Phase (Ph) Ia/Ib study evaluating zongertinib in pts with HER2-mutant advanced/metastatic NSCLC in Ph Ib. At the primary analysis (May 2024) of Ph Ib Cohort 1, the primary endpoint, confirmed response by blinded independent central review (BICR), was met in pts with pretreated HER2-mutant NSCLC within the tyrosine kinase domain (TKD). Here, we report the previously unreported median duration of response (DoR) and median progression-free survival (PFS) for Cohort 1, and first data from Cohorts 3 and 5.
Methods:
In Cohort 1, pts with pretreated HER2-mutant NSCLC within the TKD were initially randomized to receive zongertinib 120 mg or 240 mg once-daily; the 120 mg dose was selected at an interim analysis. In exploratory Cohort 3, pts had pretreated HER2-mutant NSCLC with TKD or non-TKD mutations. In Cohort 5, pts had HER2-mutant NSCLC within the TKD and were pretreated with a HER2-directed antibody-drug conjugate. In Cohorts 3 and 5, pts initially received zongertinib 240 mg. Once the 120 mg dose was selected in Cohort 1, all newly recruited pts received zongertinib 120 mg. Primary endpoint was objective response (best overall response of complete or partial response; RECIST v1.1) by BICR (Cohorts 1 and 5) and investigator review (Cohort 3). Secondary endpoints included DoR and PFS.
Results:
As of November 29, 2024, 75 pts in Cohort 1, 20 pts with non-TKD mutations in Cohort 3, and 31 pts in Cohort 5 had received zongertinib 120 mg; treatment was ongoing in 33 (44%), 4 (20%), and 13 (42%) pts, respectively. In Cohort 1, the objective response rate (ORR) by BICR was 71% (95% CI: 60-80) and disease control rate (DCR) was 96% (95% CI: 89-99). In Cohort 3, the ORR was 30% (95% CI: 15-52) and DCR was 65% (95% CI: 43-82). In Cohort 5, the ORR was 48% (95% CI: 32-65) and DCR was 97% (95% CI: 84-99). Median (95% CI) DoR and PFS were 14.1 (6.9-not evaluable [NE]) months and 12.4 (8.2-NE) months in Cohort 1 and 5.3 (2.8-NE) months and 6.8 (5.4-NE) months in Cohort 5. DoR and PFS in Cohort 3 were not yet mature. Drug-related adverse events (all/grade [G] ≥3) were reported in 97%/17%, 80%/25%, and 77%/3% of pts in Cohorts 1, 3, and 5, respectively, most commonly diarrhea in all cohorts (mainly G1). There were no cases of drug-related interstitial lung disease (ILD).
Conclusions:
Zongertinib demonstrated significant and clinically meaningful activity with a manageable safety profile in pts with pretreated advanced NSCLC harboring HER2 mutations.
[1]Heymach J,et al.Phase Ia/Ib trial of zongertinib (BI 1810631), a HER2-specific tyrosine kinase inhibitor (TKI), in patients (pts) with HER2 aberration-positive solid tumors: Updated Phase Ia data from Beamion LUNG-1, including progression-free survival (PFS) data[J]. 2024.https://ascopubs.org/doi/10.1200/JCO.2024.42.16_suppl.8514
[2]Wilding B,et al.Zongertinib (BI 1810631), an Irreversible HER2 TKI, Spares EGFR Signaling and Improves Therapeutic Response in Preclinical Models and Patients with HER2-Driven Cancers. Cancer Discov. 2025 Jan 13;15(1):119-138.https://pmc.ncbi.nlm.nih.gov/articles/PMC11726021/
|